Happy Christmas, everyone. Photo by INA FASSBENDER/AFP via Getty Images

On Saturday night, when Tier 4 was announced and imposed, I got a phone call. We hadn’t planned to take advantage of the Great Christmas Truce — it seemed mad to take our London-incubated viruses out of the M25 and give them to our retired parents. But we had planned to meet them somewhere outdoors, on neutral territory: exchange presents, say merry Christmas, perhaps drink some mulled wine out of thermos flasks. Let the children see the grandparents, let the grandparents see the children, create a thin veneer of Christmas normality.
That was suddenly impossible. So the phone call urgently said: we need to get the children’s presents to them. So I hopped into the car and rushed off to a motorway service station just outside London, where I met a grandparent, swapped a bunch of hastily wrapped gifts, paid our socially distanced respects, and set off home. What was poignant was that I saw several — five or six, probably — pairs of cars in the car park, boots open, presents ready, doing the same thing. The motorway was much busier than I would have expected on a normal Saturday night. As I was driving home a song was dedicated on the radio to a granddad who was driving from Hereford to Swansea to get his kids their presents. It was gently heartbreaking.
So I understand the anger at the new London lockdown, so late in the day, after so many Pollyannaish pronouncements and positive messages about “saving Christmas”. The new restrictions are being blamed on a new variant of SARS-Cov2 which has arisen in the South-East of England. In my more cynical moments, I had started to wonder whether they were overstating the importance of the new variant, that it was simply a convenient peg to hang the inevitable U-turn on, and I was not the only person to think that. But having spoken to some people, I have decided that was unfair.
Here’s what I’ve learned. In short: it’s real; we need to take it seriously, and watch it closely, and it will likely make life harder; but it’s not panic stations, and there’s no major reason as yet to think it will, for instance, stop vaccines from working. Also, we’ve been oddly lucky with the testing.
First: there really is a new, noticeably different strain. Ewan Birney, the deputy director-general of the European Molecular Biology Laboratory, points out that all viruses mutate, all the time. Some, such as HIV and influenza, mutate incredibly rapidly, making it very hard to create vaccines against them. SARS-Cov2 is “pedestrian” by comparison, he says, but still, new mutations arise all the time, and we don’t bother with a press conference for every one. “If we did, we wouldn’t be able to move for press conferences,” he says.
The new strain, though, really has mutated significantly more. Here is a useful chart (below), from a preprint study released over the weekend. Each blob on the chart represents one time a SARS-Cov2 virus was sequenced, so that scientists could see its genome. The higher up it is, the more it has mutated away from the original version detected in January. The further right it is, the later the date it was collected.
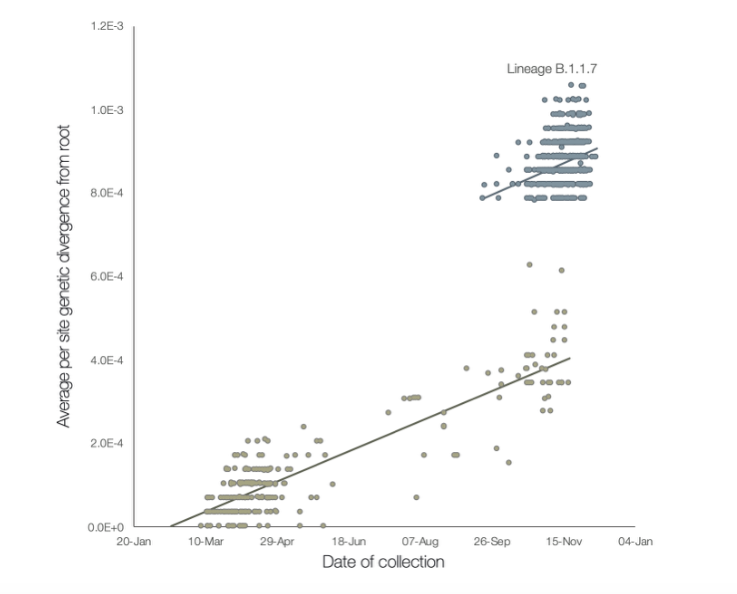
The lighter grey blobs represent the familiar SARS-Cov2 virus, slowly changing at a fairly steady, predictable rate. But the darker blobs are the new strain, B.1.1.7, which was first detected in September. You can see that although it too is changing at a steady rate, at some point before it was detected, it diverged from its parent strain significantly: it has roughly twice as many mutations per location on its genome.
Of course, we don’t care about mutations for their own sake, so it might be worth talking about what this means. A “mutation” is simply a copying error. The virus gets into your cells and commandeers them to make new copies of itself. Sometimes, as it does so, it gets it slightly wrong: random errors appear in the RNA, its equivalent to our DNA. Those errors may slightly change, for instance, the shape of a protein that it makes.
It’s wrong to think of viruses as mutating “for” something: the errors are random. Most will do nothing, or have a negative effect, like randomly adding or deleting words in a recipe. But every so often, one might do something that is useful for the virus. Perhaps it might make its protein spike a little more suited to opening up cell membranes, or help the virus evade antibodies a little more effectively. Then that version of the virus will tend to spread a bit better than the older one, and copies with that mutation will become more common in the population.
What will be helpful for the virus, of course, depends on its environment. So different viruses (or any living thing) face different “selection pressures”. For instance, if a new antiviral drug is used, then those copies of the virus which are most resistant to that drug will tend to spread better.
With viruses that have recently jumped from another species, such as SARS-Cov2, says Birney, there are usually two main pressures driving change. One is obvious: viruses that can jump more easily between hosts will tend to spread better, so viruses tend to become more efficient at transmission. Another is perhaps less so: viruses tend to become less deadly, because if your disease kills your host in two hours, you won’t have much time to get to the next one. “Viruses when they jump, usually do two things,” says Birney. “They end up mutating to transmit faster, and often to cause less disease. That’s the pathway to becoming an endemic virus.”
So the question is whether either of these things has happened in the B.1.1.7 strain, and the answer is: it’s hard to know, but probably, at least on the transmissibility point.
It’s not that you can look at a viral genome and say “Ah yes, that one’s better at spreading.” And if you notice that a particular variant is spreading faster, that may be because the variant is better at spreading, or it may just be, for instance, that it happens to be prevalent in an area where there’s a lot of transmission. There’s a lot of regional variation in what strains are where; if a new strain happens to arise in a place where, say, fewer people can work from home, then it will look like it’s good at transmission when in fact it’s just lucky. That happened early on, says Birney: the D614G mutation became the dominant one in Europe after it arose in May. It may have had a slight growth advantage, he says, although “the jury is very much still out” on that; “it’s hard to work out if it was just random chance that got it to the top or not”.
In this case, though, there are reasons to think that it probably is more effective at spreading. The New and Emerging Respiratory Virus Threats Advisory Group (NERVTAG) looked at four strands of evidence and they all centred on the B.1.1.7 strain being around 70% more effective at transmission, although there is a lot of uncertainty around that figure.
There is a theoretical reason to think it’s more transmissible, as well. The new strain had 17 “non-synonymous” mutations when it was first detected — that is, mutations which actually affect its output (there are a further six “synonymous” ones, which don’t). It’s not clear, by the way, how B.1.1.7 ended up with so many mutations. But often, multiple mutations are seen when a virus infects someone with a compromised immune system. In those patients, the virus can stay in the system for weeks and months, rather than a few days; they are often then treated with convalescent plasma. The preprint speculates that something like that may have happened here.
Birney, and Babak Javid, an immunology lab director at the University of California San Francisco, all think that is plausible, although there is no direct evidence for it. Adam Kucharski, a mathematical epidemiologist at the London School of Hygiene and Tropical Medicine, draws an analogy with antibiotic resistance: if you nuke a microbe with a full course of antibiotics, it will just die, so it won’t mutate into a resistant form. If you don’t give it antibiotics at all, it will have no pressure to mutate. But when you use antibiotics for a bit, those microbes which are a bit more resistant will tend to survive, and resistance will emerge. It’s where there are half-measures, weak selection pressures over a long time, that pathogens mutate into new forms: a virus that lives for a while in someone with a weak immune system and treated with partially effective treatments will throw out new, experimental mutations. It’s well documented.
How dangerous is the Covid mutation?
Whether or not that’s exactly what happened, B.1.1.7 definitely has several mutations, and the most notable one is a deletion: two RNA “letters” found in the main SARS-Cov2 strain have been removed.
That same mutation, 69-70del, has cropped up in a few other places. One was the Danish mink outbreak that was reported a few weeks ago; it has also been spotted in Lyon. But they are different lineages: the same mutation has happened independently. That is, says Javid, reason to think that the mutation is making the virus more transmissible. “I was sceptical,” he says, “but the same mutation has arisen in several places. If it’s random mutation it’d be less likely to keep appearing, so that’s strong evidence.” Another mutation found in B.1.17 has also cropped up in another new variant seen in South Africa.
Javid’s suspicion, and it’s only a suspicion, is that the mutations make the virus a bit harder for your immune system to kill, so it survives a little longer in the bloodstream. If you have the virus for 10% longer, then, all else being equal, you’ll be 10% more likely to pass it on. “My gut instinct is that this increases the contagious window by six hours, or 24 hours, or something,” he says. “I’m trying to think of biological ways this might result in more contagiousness, and my working hypothesis is if it’s very slightly more resistant to neutralisation by antibodies.”
It’s worth talking about testing, which is where we got a bit lucky. Testing for whether someone has a virus, using a PCR test, is usually just Y or N: the test finds the viral RNA or it doesn’t. Meanwhile, to track the spread of different variants of that virus, you have to get samples and sequence their genome, which is a longer and more involved process.
But PCR tests work by looking for a few short lengths of the viral RNA, not the whole thing. And by lucky chance, the standard PCR test looks at three different chunks, one of which happened to include the two areas removed by 69-70del mutation.
This doesn’t mean that the PCR tests don’t work. The tests look at three sites “precisely to have belt and braces” when hunting the virus, says Birney. So PCR tests which return positive results on two of the sites, but are negative on the 69-70 site, are probably telling us that the patient has B.1.1.7, not the original SARS-Cov2 strain. This means it’s been much easier to keep track of the virus. That’s why, says Birney, Patrick Vallance was able to give so much detail in his presentation of where it has spread in the UK. It’s also part of why we can make any sort of estimate about the increase in R. If we were relying on genome sequencing, even though the UK has genuinely world-leading genomic surveillance, we’d have much less info.
(The PCR tests will have to be redesigned, because the whole point is to have redundancy against mutations, and now they’ve lost some of that redundancy. But it’s been useful, nonetheless.)
So the question is: what does this mean? For our tier system, for the vaccine, for our getting back to normal? And is this strain more deadly as well as more transmissible?
For one thing, says Birney, it explains a few things about why the virus kept spreading in Kent and Essex despite them moving into Tier 3 a few weeks ago. It was working elsewhere, but not there. “This is an explanation,” he says. “Quite whether it’s the whole explanation is hard to tell.” It may mean that restrictions have to be tougher, if the new strain really does spread more easily. Essentially, Tier 3 and the new 4 are meant to bring cases down; if that’s harder to do, then we have to work harder.
For the vaccines, we can be reasonably hopeful, I think. Birney points out that there were already many strains in circulation when the vaccines were being tested; the fact that they were so effective is reason to think that they must have worked against the large majority of them. It’s not definitive — “that argument is not fully watertight,” says Birney, “because currently other strains are rare”, but it’s reassuring.
More theoretically, we can take heart from the way the immune system works. As well as B-cells which make antibodies, we have two other kinds of T-cells. One kind I think of as Stasi-like secret policemen. “They basically knock on the door of every cell and say ‘show me segments of all the proteins you have inside you’,” says Birney. “And if it finds one it hasn’t seen before, it decides it’s a virus and presses the ‘please commit suicide’ button’.”
The other kind goes up to B-cells. Each B-cell produces a unique antibody; when those antibodies destroy a pathogen, the cells keep some of chopped-up parts of those pathogens inside themselves, like prisoners, or trophies. The second kind of T-cell goes up to them and demands to see all the trophies. If it finds one it recognises, it orders the B-cell to make loads of copies of its unique antibody, stimulating a rapid immune response.
Because the T-cells only look at very short segments of protein, any single T-cell could be confused by a single mutation — if one T-cell recognises the 69-70 spot, then it wouldn’t pick up the B.1.1.7 variant with the 69-70del mutation. But because there are millions of T-cells, each remembering a slightly different bit of a protein, it’s unlikely that even quite a few mutations would remove T-cell immunity. We normally measure immunity by looking at B-cells, because they’re easier to test for, which is partly why immunity looks like it drops off quickly in Covid patients.
But T-cell immunity is longer lasting. Javid thinks it’s quite likely that the new strain could have a slightly shorter period of B-cell immunity, because of his hypothesis about resistance to antibodies, but it’s very unlikely that we’d lose T-cell immunity altogether. Kucharski adds that, in general, while the flu evolves so rapidly that antibody resistance fades in months, with seasonal coronaviruses that process takes years. Hopefully that will be true of SARS-Cov2 as well.
(There’s the added point that the new mRNA vaccines are much more plug-and-play than older ones, so in theory, you could make new vaccines for new strains pretty quickly. But I don’t know how quickly you could get new safety approval from regulators.)
Of course, this is all speculation: we need evidence, rapidly. That’s why surveillance is absolutely vital. If some new strain does outwit the vaccine, we need to know about it immediately.
Two last points. One, there’s the question of whether the new strain is more deadly, or less deadly. And the evidence is weak either way; as I said at the beginning, often you get selection pressure for viruses to become less virulent, but in the case of Covid-19 it’s in an awful sweet spot where it isn’t very deadly for most people and can spread via presymptomatic and asymptomatic people, so the selection pressure isn’t great, but it’s still very deadly among vulnerable people.
Birney says that “hospitalisation rates in the South-East [where the new strain is widespread] are not a million miles away from what you’d expect given the infection rate”, so at the moment we can only say that it’s probably not loads more or loads less dangerous. Javid says that one mutation is similar to one found in a Singapore strain which seemed to cause less deadly disease in animal trials, but again, he says, that’s speculative.
And secondly, the immediate reaction from our European neighbours has, understandably, been to close their borders with the UK. Kucharski makes the point, though, that there is a good chance that they’re shutting a stable door on a long-bolted horse. Britain has the best genetic surveillance of disease in the world, as measured in viral genomes sequenced per capita; that’s not a boast, it’s simply the case. It is very possible that the new strain is spreading rapidly in France, the Netherlands, Belgium and so on, but the surveillance is less effective at spotting it. It’s another reason why we need to improve surveillance everywhere.
At the beginning, I said I was wrong to be cynical, and to think that the new strain was being over-egged to hide the government’s own mistakes. It’s a real new strain, it seems to be more transmissible, and although it doesn’t change anything fundamental, it does make life harder. Javid describes it as an “amber light”.
But I do think there is some blame to be attached here. It might not have been obvious that things would go wrong in this way, but it was surely very possible that things could go wrong in some way. I think that it was ridiculous to think back in November that we could give any confident message about Christmas being opened up. It seems mad that millions of families have been told they can meet up, and then, five days before with the turkey already ordered, told they can’t. A more responsible attitude might have been to be much more cautious early on, to say that an open Christmas was an aspiration rather than promise.
But perhaps I’m still just sad about all those open car boots, all those presents passed from grandparent to child via an intermediary after another small hope was raised and dashed. There have been an awful lot of sacrifices, but that little indignity felt like it could have been avoided.










Join the discussion
Join like minded readers that support our journalism by becoming a paid subscriber
To join the discussion in the comments, become a paid subscriber.
Join like minded readers that support our journalism, read unlimited articles and enjoy other subscriber-only benefits.
Subscribe